Protein, Amino Acid, Essential Amino Acid, Nonessential Amino Acid, Apoprotein, Apoenzyme, Holoprotein, Zymogen, Histidine, Isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Threonine, Tryptophan, Valine, Cysteine, Arginine, Glutamine, Glycine, Proline, Alanine, Aspartic Acid, Aspartate, Glutamate, Glutamic Acid, Ornithine, Asparagine, Serine, Tyrosine
- Definitions
- Amino Acids
- Twenty common Amino Acids occur in humans, in which 9 are essential (must be ingested)
- Peptides
- Short chains of Amino Acids (two or more)
- Proteins (polypeptides)
- Long chains of peptides (which in turn are chains of Amino Acids)
- Physiology
- Structure
- Primary Protein Structure
- Specific Amino Acid sequence defining a Protein
- Secondary Protein Structure
- Regular local structure of the Protein
- Common secondary structures include alpha-helix (right-handed spiral) and beta strand (straight segment)
- Tertiary Protein Structure
- Overall spatial arrangement of a Protein (globular, membane, fibrous)
- Globular Proteins (spherical) include Hemoglobin, albumin and most enzymes
- Fibrous Proteins with straight chains include structural Proteins (e.g. Collagen, elastin, Fibrinogen)
- Quaternary Protein Structure
- Assembly of multiple Proteins combined (e.g. interwoven Collagen fibers)
- Physiology
- Function
- Functioning Protein (Holoprotein) is composed of two components
- Apoprotein
- Active Protein without Cofactors (e.g. globin in Hemoglobin)
- Apoenzyme refers to the Apoproteins of enzymes
- Apolipoprotein refers to Apoproteins of Lipoproteins (e.g. HDL, LDL)
- Prosthetic Group
- Non-Protein Cofactor (e.g. heme in Hemoglobin)
- Coenzymes refers to the prosthetic groups of enzymes
- Apoprotein
- Zymogen
- Inactive precursor to an active enzyme
- Activated by a metabolic pathway that modifies the Zymogen (e.g. cleavage, phosphorylation)
- Examples
- Gastrointestinal Metabolism (e.g. Trypsinogen to trypsin, Pepsinogen to Pepsin)
- Clotting Pathway (e.g. Prothrombin to Thrombin, Fibrinogen to Fibrin)
- Physiology
- Essential and Non-Essential Amino Acids
- Essential Amino Acids are generated by complex biosynthetic pathways not available in humans
- Histidine
- Isoleucine
- Leucine
- Lysine
- Methionine
- Phenylalanine
- Threonine
- Tryptophan
- Valine
- Conditional Essential Amino Acids are deficient in early childhood and at times of physical stress
- Arginine
- Cysteine
- Glutamine
- Tyrosine
- Glycine
- Ornithine
- Proline
- Serine
- Nonessential Amino Acids
- Includes the 8 conditional essential acids (except at times of physical stress)
- Alanine
- Aspartate
- Glutamate
- Physiology
- Amino Acid Polarity by R-Group
-
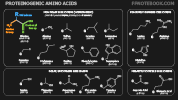
- Non-polar side chains (hydrophobic)
- Glycine
- Alanine
- Valine
- Leucine
- Isoleucine
- Cysteine
- Methionine
- Proline
- Phenylalanine
- Tryptophan
- Polar uncharged side chains
- Serine
- Threonine
- Asparagine
- Glutamine
- Tyrosine
- Positively charged side chains
- Lysine
- Arginine
- Histidine
- Negatively charged side chains
- Aspartic Acid
- Glutamic Acid
- Physiology
- Amino Acid Structures by R-Group
- Background
- Amino Acids vary by their R group
- Amino Acids share 3 common bindings to a core carbon
- Amino group or NH3 (in Proteins, binds to the COOH on the prior Amino Acid on the left)
- Hydrogen
- COOH (in Proteins, binds to the NH3 on the next Amino Acid on the right)
- Aliphatic R Group (open carbon chains)
- Glycine
- Alanine
- Valine
- Leucine
- Isoleucine
- Aromatic R Group (benzene ring)
- Tyrosine (also contains a hydroxyl group)
- Phenylalanine
- Tryptophan
- Hydroxyl Containing R Group
- Serine
- Threonine
- Sulfur Containing R Group
- Cysteine
- Methionine
- Carbonyl Containing R Group (C=O)
- Aspartate
- Asparagine
- Glutamate
- Glutamine
- Alkaline R Group
- Arginine
- Lysine
- Histidine
- Imino Acid
- Proline
- Physiology
- Protein Metabolism
-
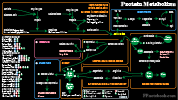
- See Protein Metabolism
- Non-Essential Amino Acid Synthesis (via Kreb Cycle or from other Amino Acids)
- Aspartate is synthesized from oxaloacetate
- Asparagine is synthesized from Aspartate
- Glutamate is synthesized from alpha-ketoglutarate
- Glutamine and Proline are synthesized from Glutamate
- Glycine and Cysteine are synthesized from Serine
- Tyrosine is synthesized from Phenylalanine
- Aspartate is synthesized from oxaloacetate
- Synthesis of other molecules from Amino Acids
- Nucleic Acids (DNA, RNA)
- Ribose 5-P backbone is synthesized from Glucose-6P or Glyceraldehyde
- Purines (ATP and GTP) are synthesized from Aspartate, Glutamine and Glycine
- Pyrimidines (TTP, CTP and UTP) are synthesized from Aspartate and Glutamine
- Sphingolipids (e.g. Sphingomyelin, Cerebroside, Ganglioside)
- Serine acts as a backbone for Fatty Acid attachment (similar to Glycerol in Triglycerides)
- Hormonal synthesis
- Histidine is converted to Histamine
- Tyrosine is converted to Thyroxine,Triiodothyronine, Melanin, Norepinephrine, Dopamine and Epinephrine
- Tryptophan is converted to Serotonin and Melatonin
- Nucleic Acids (DNA, RNA)
- Catabolism: Protein as Fuel
- Protein yields 4 kcals/g with catabolism
- Protein is the last to be catabolized in starvation (after Carbohydrate and fat)
- Proteins are broken down to Amino Acids and some small peptides before absorption
- See Gastrointestinal Metabolism
- Mediated by Stomach acid, pepsin, trypsin and peptidases
- Amino Acids are broken down into ammonia (NH3) and a carbon skeleton
- Ammonia is excreted as urea (via urea cycle)
- 2-Ketoglutarate mediated transamination removes NH3, and forms Glutamate
- Glutamate mediated oxidative deamination releases NH3 and forms 2-Ketoglutarate
- Glutamate (and asparate) donate NH3 for various synthesis (e.g. Purines, Pyrimidines)
- Ammonia enters urea cycle as carbamoyl phosphate and is ultimately excreted as urea
- Urea cycle primarily occurs in the liver
- Carbon skeleton is further processed
- Kreb Cycle (glucogenic Amino Acids)
- Acetyl CoA and Acetoacetate (ketogenic Amino Acids)
- Ammonia is excreted as urea (via urea cycle)
- Most Amino Acids are glucogenic (may generate Glucose via Gluconeogenesis)
- Exceptions: Two Amino Acids are only ketogenic (metabolized to acetyl CoA and Acetoacetate)
- Lysine
- Leucine
- Some Amino Acids are both glucogenic and ketogenic
- Isoleucine
- Tyrosine
- Phenylalanine
- Tryptophan
- Threonine
- Exceptions: Two Amino Acids are only ketogenic (metabolized to acetyl CoA and Acetoacetate)
- Amino Acids enter the Kreb Cycle at various points (for energy generation)
- Pyruvate
- Glycine
- Alanine
- Tryptophan
- Serine
- Threonine
- Cysteine
- Acetyl CoA (Ketogenic Amino Acids)
- Lysine
- Leucine
- Isoleucine
- Tyrosine
- Phenylalanine
- Tryptophan
- Threonine
- Alpha Ketoglutarate (2-Ketoglutarate)
- Proline
- Glutamate
- Glutamine
- Arginine
- Histidine
- Succinate
- Valine
- Isoleucine
- Threonine
- Methionine
- Fumarate
- Tyrosine
- Phenylalanine
- Aspartate
- Oxaloacetate
- Aspartate
- Asparagine
- Pyruvate
- Physiology
- Protein Function
- Transport and storage Proteins
- Hemoglobin And Myoglobin (oxygen transport)
- Transferrin and Ferritin (iron transport and storage respectively)
- Thyroglobulin (Thyroxine)
- Albumin (binds many molecules in plasma)
- Structural Proteins
- Collagen (cartilage, bone, connective tissue)
- Elastin (connective tissue elasticity)
- Keratin (nails, hair)
- Motion
- Myosin and actin (Muscle)
- Tubulin (cilia, flagellae)
- Hormones
- Enzymes
- Nearly all cellular metabolic processes are catalyzed by enzymes
- Miscellaneous
- Transcription factors (regulate gene expression)
- Antibody
- Clotting Factors
- Growth Factors
- References
- Goldberg (2001) Clinical Biochemistry, Medmaster, Miami, p. 24-9